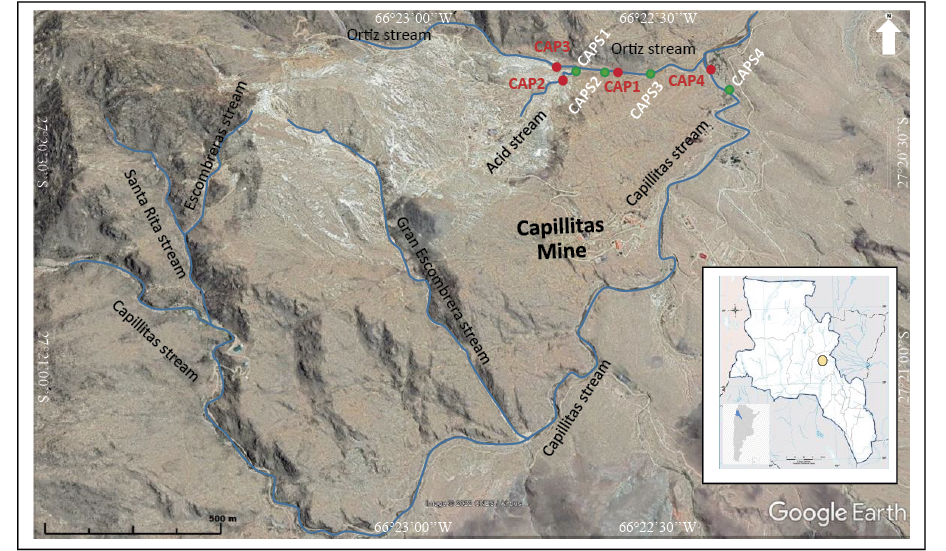
Figure 1. Satellite image of the study area with the location of sampling stations. Samples of streambed sediments: green points, water samples: red points.
STUDY AREA
Capillitas Mine is an epithermal precious and base metal vein deposit, located along the western slope of the Sierra de Capillitas, in Catamarca province (Argentina). This polymetallic epithermal deposit is part of the Farallón Negro Volcanic Complex (FNVC) that comprises numerous veins hosted in volcanic rocks and granites (Márquez-Zavalía, 2002; Putz et al., 2009). Putz et al. (2009) studied the mineralogy of this deposit and distinguished high and intermediate sulfuration events overprinted by supergene alteration. The main primary ore minerals are: pyrite (FeS2), sphalerite (ZnS), galena (PbS), chalcopyrite (CuFeS2), marcasite (FeS2), pyrrhotite (Fe7S8), tennantite (Cu6Cu4(Fe2+,Zn)2As4S12S), enargite (Cu3AsS4), bornite (Cu5FeS4), stannite (Cu2FeSnS4) and gold (Márquez-Zavalía, 2002). Rhodochrosite and quartz are the predominant gangue minerals.
The Sierra de Capillitas belongs to the Northwestern Sierras Pampeanas morphotectonic region (Ramos, 1999). The basement of the area is represented by metamorphic and plutonic rocks of Proterozoic to Lower Paleozoic age. This basement is unconformably overly by continental conglomerates, sandstones and claystones deposited in fluvial environments corresponding to the Hualfín Formation (Muruaga, 1998; 2001a, b; Bossi et al., 1999). In the study area this unit is unconformably overly by red sandstone and pelites deposited in fluvial environments assigned to the Morterito Formation (Turner, 1973) middle to upper Miocene in age (Márquez-Zavalía, 2006). The FNVC is composed of volcanic, volcaniclastic and hypabyssal rocks from upper Miocene to lower Pliocene, that unconformably overly the older units (Llambías 1972; Sasso, 1997; Sasso and Clark, 1998). In the Capillitas area the FNVC developed a diatreme filled by fragmented volcanic rocks that were later intruded by a volcanic dome (Morro Pan de Azúcar, Márquez-Zavalía, 1988, 1999). Seggiaro et al. (2014) distinguished three main volcanic facies assemblages in the FNVC: volcanic rocks ranging in composition from basalt to rhyolite, reworked volcaniclastic rocks and intrusive hypabyssal rocks.
The study area is located to the NE of the Capillitas Mine, where two streams, Ortíz and its unnamed affluent, drain spoil heaps and tailings dams that contain materials produced by old mining operations, that took place between 1855 and 1930, and recent mining activity. The Ortíz stream is an affluent of the Capillitas stream that discharges in Campo del Arenal. The Capillitas stream originates in an unmineralized area with no mining activities. However, along its upper course, it receives the water from the Santa Rita stream, which originates near the Rhodocrosite Mine and, in turn, has the Escombreras stream as a tributary. According to Yaciuk et al. (2022) the Escombreras stream, which flows through a sulfide mine dump, exhibits a pH ranging between 2.26 and 2.70 and therefore they classified it as an acid mine drainage. After its confluence with the Escombreras stream the pH of the Santa Rita stream decreases from 5.83 to 3.09 in a few meters (Yaciuk et al., 2022).
The climate of the study area corresponds to a height desert with annual average precipitation < 200 mm as the region lies in the rain shadow of the Sierra del Aconquija. On a regional scale, rainfall has a monsoon regime characterized by wet summers and dry winters, heavier rains are very frequent during southern summers (December to March) (Banchig et al., 2009). Yaciuk et al. (2022) indicated that even though the hydrological balance of the Capillitas region is markedly negative, several low-flow (0.05–14.5 L·s-1) streams exist.
MATERIAL AND METHODS
During the sampling campaign, carried out in January of 2018, four samples of water and four samples of streambed sediments were collected in a small area (~0.8 km2) downstream the Capillitas Mine (Figure 1). Water sample CAP2 corresponds to the unnamed stream, a tributary of the Ortíz stream, and sample CAP3 to the Ortíz stream; these samples were collected ~0.5 km downstream the active mining operations of Capillitas Mine, just before their confluence. The amount of suspended sediments is insignificant in both streams. Sample CAP1 was taken in a small pond situated a few meters above the riverbed level of the Ortíz stream and 100 m to the NE of its confluence with its unnamed tributary. This pond receives a low shallow groundwater flow from the NW, presumably infiltrated through the Ortiz stream bed. At the same time, it discharges into the Ortiz stream through a narrow short channel on its eastern border (Figure 2d). This pond has a very thin layer of turquoise-colored mineral precipitates at the bottom, and thus it will be called ‘turquoise pond’ hereafter (Figure 2e). Water sample CAP4 corresponds to the Capillitas stream, and it was taken a few meters before its confluence with the Ortíz stream (Figure 2f). During the sampling campaign, the flow rate of the Ortíz and the unnamed streams was several times greater than that of the Capillitas stream, where water flowed from a small spring coming from the bottom of the dry creek.
Electrical conductivity (EC), pH, and temperature were determined in situ in each sampling point employing a portable multi meter Lutron with calibrated probes. Each water sample was collected in two 200mL PVC bottles, one to determine anions and the other one to analyze major and minor cations. All the water samples were vacuum-filtered using 0.45-cellulose nitrate filters, whereas the sub samples for cations determination were acidified with nitric acid until pH < 2, to prevent precipitation during the transport to the laboratory. All the samples were stored at 4 °C until analysis. Alkalinity was determined by titration with 0.02 M HCl and phenolphthalein-bromocresol green-methyl red as colorimetric indicators, while the other major anions (SO42-, Cl-, F-) were determined by ion chromatography at the Department of Geology Chemical Laboratory (FCEyN, UBA) employing a Thermo Scientific Dionex Aquion, using the pre-column AG18 4-microns and the column AS18-4 microns, and KOH 35 mM as eluent. The concentration analysis of dissolved major cations and trace elements (Na, K, Ca, Mg, Mn, As, Cd, Zn, Cu, Fe, Al, Se, Sr, Cr, Ni, Mo, Pb and Ba) was performed on the acidified samples by Inductively Coupled Plasma Optic Emission Spectrometry (ICP-OES) PERKIN ELMER OPTIMA-8300 in the chemical laboratory of the ‘Servicio Geológico Minero Argentino’ (SEGEMAR). Certified standards supplied by NWRI – CANADA were employed for calibration. Method detection levels (M.D.L.) and Practical quantification levels (P.Q.L.) informed for the chemical laboratory were: 0.41–1.2 mg·L-1 for Na+, 0.38–1.1 mg·L-1 for K, 0.45–1.3 mg·L-1 for Ca2+, 0.24–0.74 mg·L-1 for Mg2+, 1.3–4 mg·L-1 for Mn, 2–10 µg·L-1 for As, 0.8-2.4 µg·L-1 for Cd, 0.2–0.7 µg·L-1 for Zn, 1.2–3.6 µg·L-1 for Cu, 0.4–1.1 µg·L-1 for Fe, 0.4–1.1 µg·L-1 for Al, 3–9 µg·L-1 for Se, 10–25 µg·L-1 for Sr, 0.5–1.5 µg·L-1 for Cr, 0.3–0.9 µg·L-1 for N, 0.4–1.2 µg·L-1 for Mo, 2.5–7.5 µg·L-1 for Pb and 0.8–2.4 µg·L-1 for Ba.
Active stream sediments in contact with stream water were collected in plastic bags. Samples were taken from the first centimeter of the riverbed surface, avoiding deeper sediments because they show significant changes in color and texture compared to the superficial material. CAPS1 was taken from the bed of the Ortíz stream just at the confluence with the unnamed stream, whereas CAPS2 and CAPS3 were taken from the Ortíz creek bed, respectively, ~80 and 250 m downstream from its confluence with the Acid stream (Figure 1). Sample CAPS4 corresponds to the streambed sediments of the Capillitas stream. The four sediment samples were milled in a Herzog tungsten carbide grinder and then the material <150 µm was subjected to total acid digestion in a microwave oven based on the EPA 3052 method (0.5 g is digested in 9 ml of HNO3 + 3 ml of HF + 2 ml of HCl at 180 °C for 9.5 min). The contents of Cu, Zn, As, Se, Mo, Cd, Sb, Pb, Bi, Sn and W in the extracted sediments were determined by ICP-OES PERKIN ELMER OPTIMA-8300 in the chemical laboratory of the ‘Servicio Geológico Minero Argentino’ (SEGEMAR). The contents of Al, Fe and Mn were determined by X-ray fluorescence (EDFRX-SHIMADZU) on sub samples not submitted to digestion. Certified standards supplied by NWRI – CANADA were employed for calibration.
Figure 2. Field photographs of the study area. a) View of the Ortíz stream valley upstream its confluence with the Capillitas stream. b) Longitudinal channel parallel to the Ortíz stream that would have been used for the processing of the copper ore until 1930. c) Ochre deposits and hardpan crust at the northern bank of the Ortíz stream. d) Discharge of the ‘turquoise pond’ into the Ortíz stream. e) View of the small pond containing a thin layer of turquoise-colored mineral precipitates at its bottom. The narrow discharge channel situated on its E border can also be observed. f) View of the valley of the Capillitas stream before its confluence with the Ortíz stream.
RESULTS
Hydro geochemistry
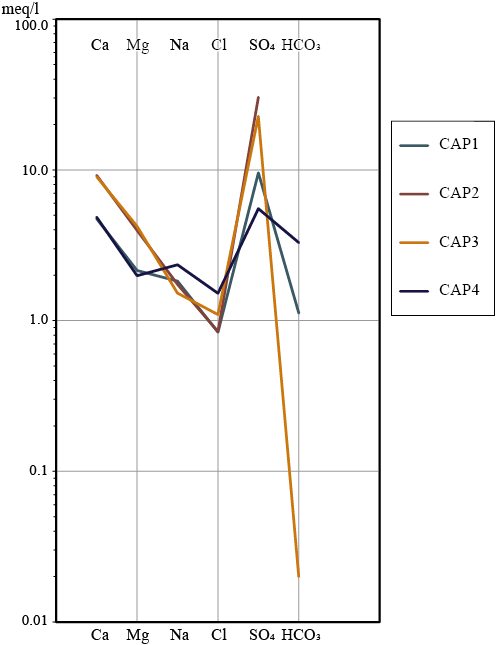
Physicochemical results are summarized in Table 1, where reference values for protection of aquatic life (PAL, Law N° 24051 Appendix IV, Argentina), and irrigation water (IW: Law N° 24051 Appendix IV, Argentina) are also included. According to the major ions concentration, the four samples correspond to calcium SO4 waters; however their pH, conductivity and contents of major (Figure 3) and minor cations are markedly different. The waters of the Ortíz stream and its unnamed tributary are acidic and depict high conductivities and high SO4 contents. The unnamed stream (CAP2) depicts pH ~3 and high contents of As (30 µg·L-1), Mn (104500 µg·L-1), Al (18698 µg·L-1) and heavy metals (Cd: 378 µg·L-1, Zn: 53790 µg·L-1 Cu: 11110 µg·L-1, Cr: 32 µg·L-1, Ni: 118 µg·L-1, Pb: 427 µg·L-1); thus it constitutes an AMD, so it will be designated as the ‘Acid stream’ hereafter. This stream also shows the highest content of Fe (14500 µg·L-1), what is consistent with the high solubility of Fe(III) under low pH oxidant waters (Brookins 1988). The Ortíz stream (CAP3) depicts pH ∼5, slightly lower EC and extremely lower concentration of Fe than the Acid stream. The lower Fe content is accordant with the Fe(III) insolubility in moderately acidic and well-oxygenated waters (Brookins, 1988). Despite its higher pH, the Ortíz stream also shows high contents of Al, Mn, Cd (738 µg L-1), Zn (52410 µg·L-1), Cu (7848 µg·L-1), Cr (16 µg·L-1), Ni (127 µg·L-1) and Pb (1897 µg·L-1).
The Capillitas stream and the turquoise pond (CAP4 and CAP1) are slightly alkaline; present lower conductivities and higher contents of bicarbonate. The turquoise pond shows concentrations of SO4 (458.8 mg·L-1), Cd (96 µg·L-1), Zn (19310 µg·L-1), Cu (215 µg·L-1), Fe (37 µg·L-1), Mn (29580 µg·L-1), Al (120 µg·L-1), Cr (7.5 µg·L-1), Ni (36 µg·L-1) and Pb (30 µg·L-1) substantially lower than the nearby Ortíz stream, whereas the As concentration was bellow the detection limit. The low content of metals in these waters is in accordance with the occurrence of a thin layer of secondary Cu-bearing sulfates precipitates at the bottom of the pond, probably associated with other metal-bearing phases (Figure 2e).
The pond is located on the hillside, above the riverbed level of the Ortíz stream, consequently the slight alkalinity of this water is probably caused by dilution with locally infiltrated rainwater that was not in contact with mine wastes. Therefore, the precipitation of the newly formed phases should be mainly driven by the pH increase.
The waters of the Capillitas stream (CAP4) are alkaline and show lower EC and the lowest contents of Al (63 µg·L-1), Mn (105 µg·L-1), Zn (115 µg·L-1), Cu (12 µg·L-1), Cr (1.5 µg·L-1), and Pb (8.7 µg·L-1). Whereas Ni and Cd were bellow detection limits the concentrations of Mn, Zn, Cu and Pb at this sampling point exceed the limiting values for the protection of aquatic life in Argentine (Table 1). On the contrary, it shows a slightly higher content of As (17 µg·L-1) than the Ortíz stream. Upstream its confluence with the Ortíz stream the waters of the Capillitas stream are suitable for irrigation according to the environmental legislation of Argentina. This might seem unlikely at first because a few kilometers upstream of the sampling point, the Capillitas stream receives the waters from Santa Rita stream, which has a small mine at its source (Santa Rita Mine) and a tributary that originates in a sulfide mine and flows through the mineralized area. However, a recent study of the upper and middle sections of the Capillitas basin has found that, in the area of Santa Rita Mine, the acidity arising from pyrite oxidation is partially neutralized by the weathering of ubiquitous rhodochrosite (Yaciuk et al., 2022).
The waters of the Acid and Ortíz streams and the turquoise pond show concentrations of Al, Mn, Cd, Zn, Cu, and Pb that exceed one hundred to five thousand times the limiting concentrations for the protection of aquatic life and from five to sixteen times in the cases of Cr and Ni. Moreover, the concentrations of Mn, Cd, Zn, Cu, and Pb of these watercourses also exceed, in different degrees (2 to 2900) the allowable maximum values for irrigation waters (Table 1).
In order to rank the pollution degree of the studied watercourses we employed the environmental risk factor proposed by Riba et al. (2002) for sediments that was later extended by Sarmiento et al. (2011) to waters. This factor was defined as,
ERF= (Cn – CSQV)/CSQV
Where Cn is the concentration of the metal in water and CSQV is the highest concentration measure of an element in the sampling site, not related with biological effects. In order to apply the ERF to waters, CSQV was replaced by the continuous concentration criteria (CCC), estimated by the Environmental Protection Agency (USEPA), following Sarmiento et al. (2011). According to the USEPA definition, CCC represent the highest concentration of an element in surface water to which an aquatic community can be indefinitely exposed without resulting in unacceptable effects (US-EPA 2002). This implies that positive ERF values indicate environmental risk, specifically for aquatic life. The ERF was calculated for As, Cd, Zn, Cu, Cr, Ni and Pb (Table 2). CCC values were not included in US-EPA (2002) for the other determined metals. The ERF obtained for the Acid and Ortíz streams and for the turquoise pond indicates that Cd, Cu, Zn and Pb represent a considerable environmental risk for these watercourses. Namely, an ERF from 383 to 2951 was obtained for Cd, from 23 to 1233 for Cu, from 160 to 447 for Zn, and from 11 to 758 for Pb. In the case of Cr and Ni, they only represent a risk in the Ortíz and Acid streams; although the ERF values are considerably lower (0.45 to 1.92) than those calculated for the other heavy metals. On the contrary, As does not represent an environmental risk in these watercourses according with this criterion. Regarding the Capillitas stream, only Pb and minimally Cu represent an environmental risk according to the computed ERF (Table 2).
|
|
M.D.L.
|
P.Q.L.
|
CAPS1
|
CAPS2
|
CAPS3
|
CAPS4
|
PEL
|
Bn
|
|
Al
|
%
|
0.04
|
0.12
|
5.2
|
5.6
|
6.7
|
6.9
|
|
|
|
As
|
µg·g-1
|
0.4
|
1.2
|
851
|
616
|
1291
|
17
|
17
|
41.81
|
|
Bi
|
µg·g-1
|
1
|
2
|
<2
|
<2
|
<2
|
17
|
|
|
|
Cd
|
µg·g-1
|
0.16
|
0.5
|
79
|
36
|
40
|
6.5
|
3.5
|
0.34
|
|
Zn
|
µg·g-1
|
0.04
|
0.14
|
26031
|
38768
|
15722
|
206
|
315
|
119.78
|
|
Cu
|
µg·g-1
|
0.24
|
0.72
|
806
|
5737
|
7802
|
130
|
197
|
23.05
|
|
Sn
|
µg·g-1
|
2
|
4
|
38
|
69
|
54
|
36
|
|
|
|
Fe
|
%
|
0.03
|
0.08
|
4.7
|
3.1
|
5.5
|
2.7
|
|
|
|
Mn
|
%
|
0.06
|
0.19
|
1.4
|
1.2
|
2.6
|
0.1
|
|
0.09
|
|
Mo
|
µg·g-1
|
0.08
|
0.24
|
1.7
|
0.46
|
<0.24
|
13
|
|
|
|
Pb
|
µg·g-1
|
0.2
|
1.5
|
4515
|
3268
|
4596
|
75
|
91.3
|
27.7
|
|
Se
|
µg·g-1
|
0.6
|
1.8
|
< 1.8
|
< 1.8
|
< 1.8
|
< 1.8
|
|
|
Figure 4. a) Orange-colored sediment of the Ortíz stream at the point of its confluence with the Acid stream (CAPS1). b) and c) Ochre-colored streambed sediments taken ~80 (CAPS2) and 250 m (CAPS3) downstream of CAPS1 in the Ortíz creek. d) Light brown-colored sediment of Capillitas stream (CAPS4) before its confluence with the Ortíz stream.
DISCUSSION
The Acid stream that drains the mineralized area and the stockpile of the Capillitas Mine constitutes an AMD as it shows pH ~3, high EC, high contents of SO4, As and heavy metals. However, in comparison with the nearby Escombreras stream, that also represents an AMD (Yaciuk et al., 2022), it presents significantly lower contents of SO4, Cu, Fe, Zn and Cd, and a slightly higher pH. On the contrary, the Acid stream shows higher contents of Pb than the Escombreras stream. These differences could be due to the fact that the Escombreras stream was sampled in close proximity to a sulfide mine dump of large dimensions comprising chalcopyrite, sphalerite, galena and pyrite (Yaciuk et al., 2022). Moreover, the lower pH of the Escombreras stream, as well as its higher EC, indicate stronger dissolution, lixiviation and metal mobility processes. Furthermore, the higher content of Pb in the Acid stream could be due to several reasons: 1) a higher proportion of galena in the stockpile that is drained by the Acid stream; b) Pb could have been removed from the solution by co-precipitation or adsorption onto the metal sulfates that appear as efflorescent salts along the course of the Escombreras stream (Yaciuk et al., 2022).
The Ortíz stream shows a higher pH and lower EC than the Acid stream; but higher concentrations of Cd, Ni and Pb. Heavy metals should mainly derive from alteration of metal sulfides, mainly galena and sphalerite, occurring in the tailings dam that the Ortíz stream crosses. In relation to Cd, Yaciuk et al. (2022) found that sphalerite is the main carrier of this element in the veins of the sulfide mine that crosses the Escombreras stream. Besides, a minor contribution arising from dissolution of metal bearing sulfurs of the mineralized area to the Ortíz stream could not be discarded. The differences in heavy metal and As contents between the Acid and Ortíz streams should be mainly controlled by the contrasting mineralogy of the mine dumps that they cross.
The metal content of the Ortíz and Acid streams imply a considerable environmental risk, since it is known that dissolved heavy metals such as Zn, Pb, Cd and Cu are highly toxic to many aquatic organisms (Verma and Dwivedi, 2013; Jain et al., 2016; Masindi and Muedi, 2018). The Ortíz stream also represents a potential threat to groundwater resources as significant water infiltration occurs in its channel near the confluence with the Capillitas stream, which is evidenced by a considerable reduction of flow rate.
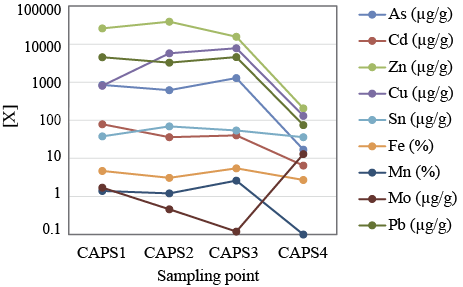
In the case of the Capillitas stream, only Pb, and minimally Cu, represent an environmental risk according to the ERF index. However, it is worth noting that the concentration of Mn, Zn, Cu Fe, Al and Pb shown by Capillitas stream before its joint with the Ortíz stream are markedly higher than those obtained around one kilometer upstream by Yaciuk et al. (2022). This suggests that the attenuation processes proven by these authors in the upper section of the Capillitas basin are counterbalanced downstream by the input of toxic elements, probably lixiviated from the mine wastes stacked along the south bank of the Ortíz creek. A specific concern is that the Capillitas stream shows similar As contents than the Ortíz stream, even though the sampling point CAP4 is located far away from the mine dumps that affected the tributaries of the Capillitas stream in its upper course. A similar case of alkaline waters showing high contents of dissolved As was reported downstream the La Soterreña mine by Larios et al. (2012). It is worth noting that in both cases As and Fe contents are not correlated, indicating that As cannot arise from dissolution of Fe(III) oxy-hydroxides. In the case of the La Soterreña mine, the high level of As in the alkaline waters was related with the dissolution of arsenates (Larios et al., 2012), Unfortunately, in the case of the Capillitas stream, there was not enough data to identify the processes controlling As mobilization.It is interesting to compare the contents of heavy metals between the waters of the Acid stream with those of the streambed sediments at the confluence with the Ortíz stream (CAPS1). The water shows contents of Cd, Zn, Cu and Mn markedly higher than those of the streambed sediments, indicating that these potentially toxic elements remain in great part dissolved. On the contrary, the contents of Fe, As, and Pb are extremely higher in the sediment than in the water. This means that the pH increase, caused by the mixing of the Acid and Ortíz streams, promotes the precipitation of authigenic phases that capture a significant part of the dissolved heavy metals and metalloids. The intense ochre color of these streambed sediments, as well as their high content of Fe and As, evidence the presence of iron oxy-hydroxides. The high affinity of As for newly-formed Fe(III) precipitates is well established and is regarded as the main attenuation mechanism of As migration (Smedley and Kinniburgh, 2002, Asta et al., 2010, Larios et al., 2012). It is known that these ochre sediments can stifle the stream bed and benthic habitat (Jain et al. 2016). Downstream the contents of heavy metals and As in the sediments of the Ortíz creek vary in a complex way. The highest content of Cd was found in CAPS1, whereas Zn and Sn show higher concentrations in CAPS2. The highest content of As, Cu, Fe, Mn and Pb was observed in the sediments taken ~250 mdownstream (CAPS3). Thus, the polluted sediments have been dispersed by water currents, probably in combination with dissolution/precipitation, adsorption and other chemical processes.
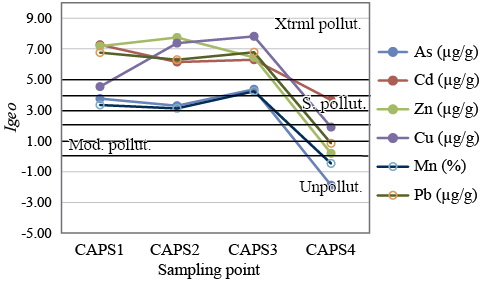
The Igeo obtained for stream sediments from the Ortíz creek indicates that the contents of Cd, Zn, Cu, Mn, Pb and As largely exceed the average values computed for the unpolluted sediments of the study area. It is well known that the total metal content in sediments is not a reliable indicator of toxicity, as contaminants can be allocated in numerous phases with different bioavailability degrees (Landrum and Robbins, 1990). However, the water in contact with these streambed sediments shows contents of Cd, Zn, Cu and Mn remarkably higher than the sediments, indicating the need for a remediation program leading to the isolation of potentially toxic metal(loids) in order to meet environmental regulations and international standards of the mining industry.
The water courses that drain materials from the Capillitas Mine, as well as their streambed sediments, show high contents of potentially toxic elements that can affect the hydrochemical water quality of the Capillitas stream, which is used by the rural population. Thus, effective actions are needed in the Capillitas Mine in order to avoid the dispersion of toxic elements into the watershed of this area.
The issues raised by the pollution related with Capillitas Mine is not unique in Argentina. In the case of NW Argentina, a study demonstrated the deleterious effect of Aguilar Mine on the water quality of Casa Grande stream and Yacoraite River, displaying high concentrations of Mo, Pb, Zn and Cd (Kirschbaum et al., 2012). La Concordia mine (Salta province, Argentina) represents another worrying case. It was closed in the mid ’80, leaving sulfide-rich residues accumulated in tailing dams in the valley of the Concordia stream, a 14 km long watercourse that is a tributary of the San Antonio River, which constitutes the main water supply of San Antonio de los Cobres, the main city of the region (~6000 inhabitants). Chemical analysis of the stream water close to the mine demonstrate the occurrence of highly acidic waters (3 < pH < 4) with concentrations of As, B, Cd, Mn, Pb, and Zn that largely exceed the guideline values for drinking water in Argentina (Kirschbaum et al., 2012). A later study demonstrates the high mobility of the As retained in secondary (hydrous)sulfate forming large saline crusts or aggregates disseminated within the matrix of the oxidized levels of La Concordia mine tailings (Nieva et al., 2016). The high concentration of As observed in a sampling site of the Capillitas stream, located far away from the mine dumps that affected its upper basin tributaries, indicates a high mobility of As, in accordance with the behavior observed in La Concordia mine tailings (Nieva et al., 2016).
Figure 5. Concentration of some toxic metals and metalloids in the streamb-ed sediments sampled along the Ortíz creek.
Figure 6. Variation of the streambed sediments Igeo along the Ortíz creek between sampling stations. Horizontal bold lines indicate the threshold values for the different categories of Igeo (Unpollut.: unpolluted, Mod. pollut: moderately polluted, strongly pollut.: S. pollut.: strongly polluted, Xtrml pollut: extremely polluted). The values of Bn employed in the Igeo calculation were the average composition of As, Cd, Zn, Cu, Mn and Pb in unpolluted streambed sediments reported in the geochemical chart of the region (Ferpozzi et al. 2003).
CONCLUSIONS
Sulfide oxidation from the tailings and spoil heaps at Capillitas Mine originated a typical acid mine drainage, rich in dissolved metal(loids) and SO4, that leaks into the Ortíz and Acid streams which cross the site. As a result, the water quality is strongly affected, as it shows acidic pH values (i.e., pH < 4) and concentrations of metals and As that highly exceed the guideline values for the protection of aquatic life and irrigation specified in the legislation of Argentina. Moreover, the high concentration of As depicted by the Capillitas stream in a sampling point located far away from the mine dumps, that affected its upper basin, evidences the high mobility of this element and represents an environmental concern. The increase of pH resulting from the mixing of the Acid stream and the Ortíz stream promotes the precipitation of Fe oxy-hydroxides that adsorb part of the dissolved As onto their surfaces and also provokes the precipitation of Pb-bearing phases, probably sulfates. The content of heavy metals and As in the sediments of the Ortíz creek varies in a complex pattern. The highest content of Cd was found where the Ortiz and Acid steams join, whereas Zn and Sn show higher concentrations in the sediments sampled ~80 m downstream. The highest content of As, Cu, Fe, Mn and Pb was observed in the sediments taken ~250 m downstream. Thus, the polluted sediments are being dispersed by water currents probably in combination with dissolution/precipitation and adsorption processes. According to the obtained Igeo, the three streambed sediment samples from the Ortíz creek are extremely polluted with Cd, Zn and Pb, and range from strongly to extremely polluted with Cu, As and Mn. Whereas the sediments from Capillitas creek are strongly polluted with Cd, moderately polluted for Cu and unpolluted to moderately polluted with Pb and Zn.
ACKNOWLEDGMENTS
We acknowledge Alejandro Diaz, Patricia Cirello and Patricia Claramunt from the Chemical Laboratory of ‘Servicio Geológico Minero Argentino’ for conducting the chemical analyses of the water and sediments samples and to Paula Taich from the Department of Geology Chemical Laboratory (FCEyN, UBA) for the anion determination in the water samples. We also thank Sofía Victoria Tarabusi for revising the English text. This research was developed under the grant PDTS 244 (Consejo Interuniversitario Nacional – Consejo Nacional de Actividades Científicas y Técnicas). Finally, we deeply thank the editorial work and comments by the editor Dra. Natalia Pardo Villaveces, and the constructive suggestions by Dr. Rodolfo Fernández-Martínez and two anonymous reviewers, which significantly improved a previous version of the manuscript.
REFERENCES
Asta, M.P., Ayora, C., Román-Ross, G., Cama, J., Acero, P., Gault, A.G., Charnock, J.M., Bardelli, F., 2010, Natural attenuation of arsenic in the Tinto Santa Rosa acid stream (Iberian Pyritic Belt, SW Spain): The role of iron precipitates: Chemical Geology, 271, 1-12.
Banchig, A.L., Moreiras, S.M., Banchig, P.N., 2009, Eventos de avalanchas y represamientos reiterados de ocurrencia prehistórica en la cuenca del río Villavil, Sierra de Aconquija, Andalgalá, Catamarca: Revista de la Asociación Geológica Argentina, 65, 805-814.
Bighman, J.M., Nordstrom, D.K., 2000, Iron and aluminum hydroxysulfates from acid sulfate waters, in Alpers C.N., Jambor J.L., Nordstrom D.K. (eds), Sulfate minerals: crystallography, geochemistry, and environmental significance. Reviews in Mineralogy and Geochemistry: Chantilly, Virginia, USA, Mineralogical Society of America, 351-403
Bossi, G.E., Muruaga, C., Gavriloff, I.J.C., 1999, Ciclo Andino. Neógeno-Pleistoceno. Sedimentación, in González Bonorino, G., Omarini, R., Viramonte, J. (eds.), Geología del Noroeste Argentino: Salta, Argentina, Universidad Nacional de Salta-CONICET, 329-360.
Brookins, D.G., 1988, Eh-pH Diagrams for Geochemistry: Heidelberg, Springer –Verlag, 232 pp.
CCME (Canadian Council of Ministers of the Environment), 2001, Canadian sediment quality guidelines for protection of aquatic life: Summary tables: Winnipeg, Environmental Quality Guidelines, Environment Canada, Guidelines division.
Delfino, D.D., Quesada, M.N., Dupuy, S.J.P., 2014, El ciclo del cobre en Minas Capillitas (provincia de Catamarca, Argentina) en la segunda mitad del siglo XIX: tensiones entre lógicas productivas, escalas tecnológicas y unidades sociales: Revista de Estudios Atacameños, 48, 119-140.
DelValls, T.A., Forja, J.M., Gómez-Parra, A., 1998, Integrative assessment of sediment quality in two littoral ecosystems from the Gulf of Cádiz, Spain: Environmental Toxicology, 17(6), 1073-1084.
Do Campo, M., Valenzuela, M.F., Ferro, L., 2020, Dispersión de contaminantes a partir de residuos mineros de una antigua planta de fundición (Ingenio Muschaca) ubicada en el distrito Choya (Andalgalá, Catamarca): Revista de la Asociación Geológica Argentina, 77, 221-230.
Fernández-Turiel, J.L., Aceñolaza, P., Medina, M.E., Llorens, J.F., Sardi, F., 2001, Assessment of a smelter impact area using surface soils and plants: Environmental Geochemistry and Health, 23, 65-78.
Ferpozzi, L., Turel, A., Vargas, D., Valladares, H., Jara, A., Siehankiewicz, D., Casa, A., 2003, Datos geoquímicos de Cu, Pb, Zn y ubicación de sitios de muestreo de corriente del Plan NOA I Geológico-Minero, Hoja 2766 III Belén, Provincia de Catamarca, República Argentina: Buenos Aires, Argentina, Instituto de Geología y Recursos Minerales, Servicio Geológico Minero Argentino, 155 pp.
Gałuszka, A., Migaszewski, Z.M., Dołęgowska, S., Michalik, A., 2018, Geochemical anomalies of trace elements in unremediated soils of Mt. Karczówka, a historic lead mining area in the city of Kielce, Poland: Science of the Total Environment, 639, 297-405.
Jain, R.K., , Z., ., 2016, Environmental Impacts of Mining, in Ravi, K.J., Zengdi “Cindy”, C., Domen, J.K. (eds.), Environmental Impact of Mining and Mineral Processing Management, Monitoring, and Auditing Strategies: Oxford, Butterworth-Heinemann, 53-157.
Jennings, S.R., Neuman, D.R., Blicker, P.S., 2008, Acid Mine Drainage and Effects on Fish Health and Ecology: A Review: Bozeman, Montana, USA, Reclamation Research Group, 26 pp. Retrieved from: http://www.pebblescience.org/pdfs/Final_Lit_Review_AMD.pdf.
Johnson, D.B., Hallberg, K.B., 2005, Acid mine drainage remediation options: a review: Science of the Total Environment: 338, 3-14.
Kirschbaum, A., Murray, J., Arnosio, M., Tonda, R., Cacciabue, L., 2012, Pasivos ambientales mineros en el noroeste de Argentina: aspectos mineralógicos, geoquímicos y consecuencias ambientales: Revista Mexicana de Ciencias Geológicas, 29, 248-264.
Kossoff, D., Hudson-Edwards, K.A., Howard, A.J., Knight, D., 2016, Industrial mining heritage and the legacy of environmental pollution in the Derbyshire Derwent catchment: Quantifying contamination at a regional scale and developing integrated strategies for management of the wider historic environment: Journal of Archaeological Science, Reports, 6, 190-199.
Landrum, P.F., Robbins, J.A., 1990, Bioavailability of sediment-associated contaminants to benthic invertebrates, in Baudo, R., Giesy, J.P., Muntau, H. (eds.), Sediments: Chemistry and toxicity of in-place pollutants: Boca Raton, FL, USA, Lewis Publishers, 237-263.
Llambías, E.J., 1972, Estructura del grupo volcánico Farallón Negro, Catamarca, República Argentina: Revista de la Asociación Geológica Argentina, 27, 161-169.
Larios, R., Fernández-Martínez, R., Silva, V., Loredo, J., Rucandio, I., 2012, Arsenic contamination and speciation in surrounding waters of three old cinnabar mines: Journal of Environmental Monitoring: 14, 531-542.
Márquez-Zavalía, M.F., 1988, Mineralogía y génesis del yacimiento Capillitas (Catamarca, República Argentina): Salta, Argentina, Universidad Nacional de Salta, Ph D Thesis, 258 pp.
Márquez-Zavalía, M.F., 1999, El yacimiento Capillitas, provincia de Catamarca, in Zappettini, E.O. (ed.), Yacimientos Minerales de la República Argentina: Buenos Aires, Argentina, Instituto de Geología y Recursos Minerales –SEGEMAR, 1643-1652.
Márquez-Zavalía M.F., 2002, Minas Capillitas, un depósito epitermal del noroeste argentino, in Trombotto, D.T., Villalba, R. (eds.), IANIGLA, 30 Años de investigación básica y aplicada en ciencias ambientales: Mendoza, Instituto Argentino de Nivología, Glaciología y Ciencias Ambientales, 249-253.
Márquez-Zavalía, M.F., 2006, El extremo rico en As de la serie jordanita-geocronita de Mina Capillitas, Catamarca: Revista de la Asociación Geológica Argentina, 61, 231-235.
Masindi, V., Muedi, K.L., 2018 , Environmental Contamination by Heavy Metals in Saleh, H.E-D.M., Aglan, R.F. (eds.), Heavy Metals: London, IntechOpen, 10.5772/intechopen.76082.
Méndez-Ramírez, M., Armienta Hernández, M.A., 2012, Distribución de Fe, Zn, Pb, Cu, Cd y As originada por residuos mineros y aguas residuales en un transecto del Río Taxco en Guerrero, México: Revista Mexicana de Ciencias Geológicas, 29(2), 450-462.
Müller, G., 1981, Die Schwermetallbelastung der sediments des Neckars und seiner Nebenflusse: eine Bestandsanfnahme: Chemiker Zeitung, 105,157-164.
Murray, J., Kirschbaum, A., Dold, B., Guimaraes, E.M., Miner, E.P., 2014, Jarosite versus soluble iron-sulfate formation and their role in acid mine drainage formation at the Pan de Azúcar mine tailings (Zn-Pb-Ag), NW Argentina: Minerals 4, 477-502, https://doi.org/10.3390/min4020477
Muruaga, C.M., 1998, Estratigrafía y sedimentología del Terciario Superior de la sierra de Hualfín, entre las localidades de Villavil y San Fernando, Provincia de Catamarca: Tucumán, Argentina, Universidad Nacional de Tucumán. Facultad de Ciencias Naturales e Instituto Miguel Lillo, PhD Thesis, 270 pp.
Muruaga, C.M., 2001a, Estratigrafía del Miembro El Jarillal (Formación Chiquimil, Mioceno superior), la Sierra de Hualfín, Catamarca: Acta Geológica Lilloana, 18, 265-280.
Muruaga, C.M., 2001b, Estratigrafía y desarrollo tectosedimentario de sedimentos terciarios en los alrededores de la Sierra de Hualfín, borde suroriental de la Puna, Catamarca, Argentina: Latin American Journal of Sedimentology and Basin Analysis, 8, 27-50.
Navarro, A., Collado, D., Carbonel, M., Sanchez, J.A., 2004, Impact of mining activities on soils in a semi-arid environment: Sierra Almagrera district, SE Spain: Environmental Geochemistry and Health, 26, 383-393.
Nieva, N.E., Borgnino, L., Locati, F., García, M.G., 2016, Mineralogical control on arsenic release during sediment–water interaction in abandoned mine wastes from the Argentina Puna: Science of the Total Environment, 550, 1141-1151.
Nordstrom, D.K., 2015, Baseline and premining geochemical characterization of mined sites: Applied Geochemistry, 57, 17-34.
Putz, H., Paar, W.H., Topa, D., 2009, A contribution to the knowledge of the mineralization at Mina Capillitas, Catamarca: Revista de la Asociación Geológica Argentina, 64, 514-524.
Rack, M.G., Nillni, A.M., Do Campo, M., Valenzuela, M.F., Ferro, L.F., 2017, Presencia de metales pesados en un suelo aledaño a una escombrera polimetálica en mina La Ferrocarrilera, Lago Fontana. Chubut, Argentina: Revista Ingeniería Sanitaria y Ambiental, Asociación Argentina de Ingeniería Sanitaria y Ciencias del Ambiente, 131, 81-85.
Ramos, V.A., 1999, Las provincias geológicas del territorio argentino in Caminos, R. (ed.), Geología Argentina: Buenos Aires, Instituto de Geología y Recursos Minerales, 41-96.
Ramos, O.E.R., Cáceres, L.F., Muñoz, M.R.O., Bhattacharya, P., Quino, I., Quintanilla, J., Sracek, O., Thunvik, R., Bundschuh, J., García, M.E., 2012, Sources and behavior of arsenic and trace elements in groundwater and surface water in the Poopó Lake Basin, Bolivian Altiplano: Environmental Earth Sciences, 66, 793-807.
Resongles, E., Casiot, C., Freydier, R., Dezileau, L., Viers, J., Elbaz-Poulichet, F., 2014, Persisting impact of historical mining activity to metal (Pb, Zn, Cd, Tl, Hg) and metal-loid (As, Sb) enrichment in sediments of the Gardon River, Southern France: Science of the Total Environment, 481, 509-521.
Responsible Mining Foundation (RMF) and Centro Vincular-PUCV, 2020, Responsible mining in Latin America and the Caribbean? Assessing how mining companies address public interest issues: RMF and Centro Vincular-PUCV, 24 pp. www.responsibleminingfoundation.org, https://www.responsibleminingfoundation.org/lac-study2020/
Riba, I., Delvalls, T.A., Forja, J.M., Gómez-Parra, A., 2002, Influence of the Aznalcóllar mining spill on the vertical distribution of heavy metals in sediments from the Guadalquivir estuary (SW Spain): Marine Pollution Bulletin, 44, 39-47.
Sarmiento, A.M., DelValls, A., Nieto, J.M., Salamanca, M.J., Caraballo, M.A., 2011, Toxicity and potential risk assessment of a river polluted by acid mine drainage in the Iberian Pyrite Belt (SW Spain): Science of the Total Environment, 409, 4763-4771.
Sasso, A.M., 1997, Geological evolution and metallogenic relationships of the Farallon Negro volcanic complex, NW Argentina: Kingston, Ontario, Canada, Queen’s University, Ph D Thesis, 842 pp.
Sasso, A.M., Clark, A.H., 1998, The Farallón Negro Group. NW Argentina: Magmatic, hydrothermal and tectonic evolution and implications for Cu-Au metallogeny in the Andean back-arc: Society of Economic Geologists Newsletter, 34, 8-18.
Seggiaro, R., Caffe, P.J., Becchio, R., Galli, C., Arnosio, M., Da Poián, G., 2014, Evolución tectónica andina entre las Sierras de Hualfín, Capillitas y extremo sur de Aconquija, Provincia de Catamarca: Revista de la Asociación Geológica Argentina, 71, 500-512.
Smedley, P.L., Kinniburgh, D.G., 2002, A review of the source, behaviour and distribution of arsenic in natural waters: Applied Geochemistry, 17, 517-568.
Stefanowicz, A.M., Wochb, M.W., Kapusta, P., 2014, Inconspicuous waste heaps left by historical Zn–Pb mining are hot spots of soil contamination: Geoderma, 235-236, 1-8, https://doi.org/10.1016/j.geoderma.2014.06.020
Turner, J.C., 1973, Descripción Geológica de la Hoja 11d Laguna Blanca (Provincia de Catamarca): Buenos Aires, Argentina, Servicio Nacional de Minería y Geología, 71 pp.
US-EPA (United States Environmental Protection Agency), 2002, National recommended water quality criteria: Washington, D.C., Environmental Protection Agency, Technical Document, EPA-822-R-02- 047, 22 pp.
Verma, R., Dwivedi, P., 2013, Heavy metal water pollution- A case study: Recent Research in Science and Technology, 5, 98-99.
Yaciuk, P.A., Colombo, F., Lecomte, K.L., De Micco, G. Bohé, A.E., 2022, Cadmium sources, mobility, and natural attenuation in contrasting environments (carbonate-rich and carbonate-poor) in the Capillitas polymetallic mineral deposit, NW Argentina: Applied Geochemistry, 136, 105152.
Younger, P.L., Banwart, S.A., Hedin, R.S., 2002, Mine water: hydrology, pollution, remediation: Environmental Pollution 5: Kluwer Academic Publishers, Dordrecht, 442 pp.