ABSTRACT
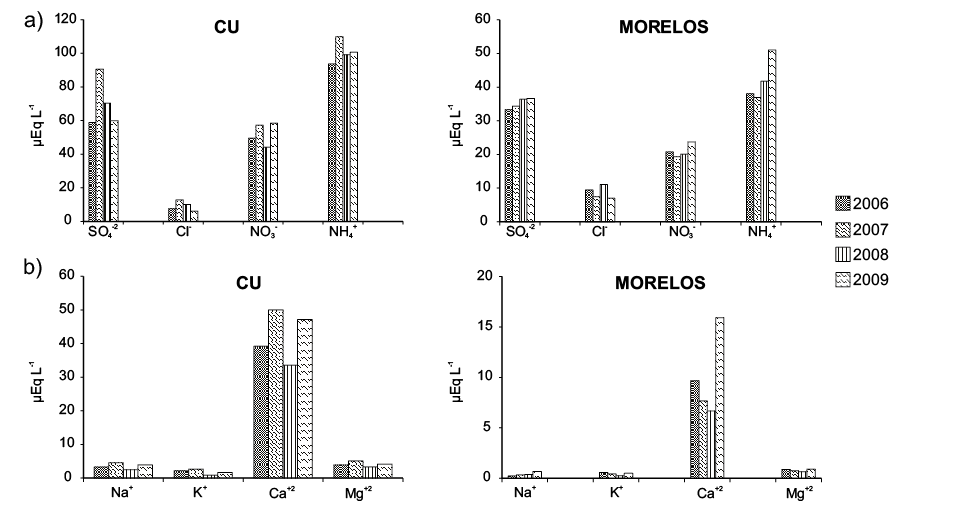
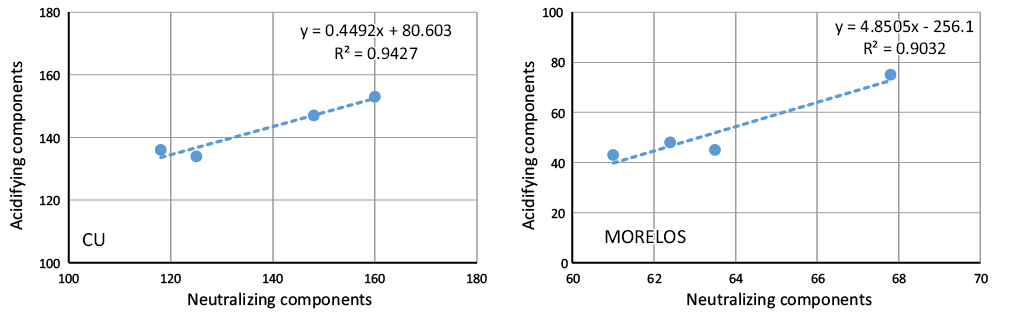
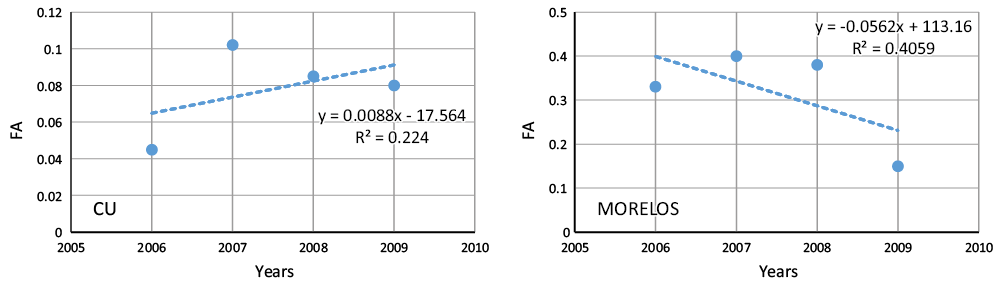
The objective of the study was to evaluate the chemical composition of rainwater in urban and suburban areas of central Mexico to identify the possible sources of rainwater contamination. The rainwater was collected at Universidad Nacional Autónoma de Mexico, Campus Ciudad Universitaria (CU), in the southern part of Mexico City at 2200 meters above sea level (m a.s.l.). CU has many green areas with high to moderate traffic densities where air quality presents serious problems of pollution by particulate matter. The other sampling site is a forested rural area (Tlalnepantla), State of Morelos, Mexico, a suburban area 86 km to the south of Mexico City. A total of 145 rainwater samples were collected in the rain period from 2006 to 2009. The ions analyzed were the following: SO42-, NO3-, Cl-, HCO3-, Na+, K+, Ca2+, Mg2+, NH4+ and H+. Ammonium was the most abundant cation in both sites and is one of those responsible for the neutralization of acidic compounds in the atmosphere. The relative abundance of the inorganic anions present in the rainwater was in the following order: SO42- > NO3- > Cl-, for the alkaline metals the order was Ca2+ >Mg2+ > Na+ > K+ and Ca2+ >Mg2+ >K+ >Na+ for CU and Morelos, respectively. A correlation analysis shows a strong positive correlation among the ions, indicating that the most important source was anthropogenic. Air mass back trajectories were associated with the SO42-, Ca2+, Mg2+, NH4+ and H+ concentrations observed on each rainy day. Four factors were used in the statistic analysis and was weighted within each factor. Weights greater than 0.5 are considered to be significant components of each factor. The four factors explain 84.7 % of the total variance of all of the data for CU and 66.9 % for Morelos. All of these factors were associated with all of the analyzed ions.
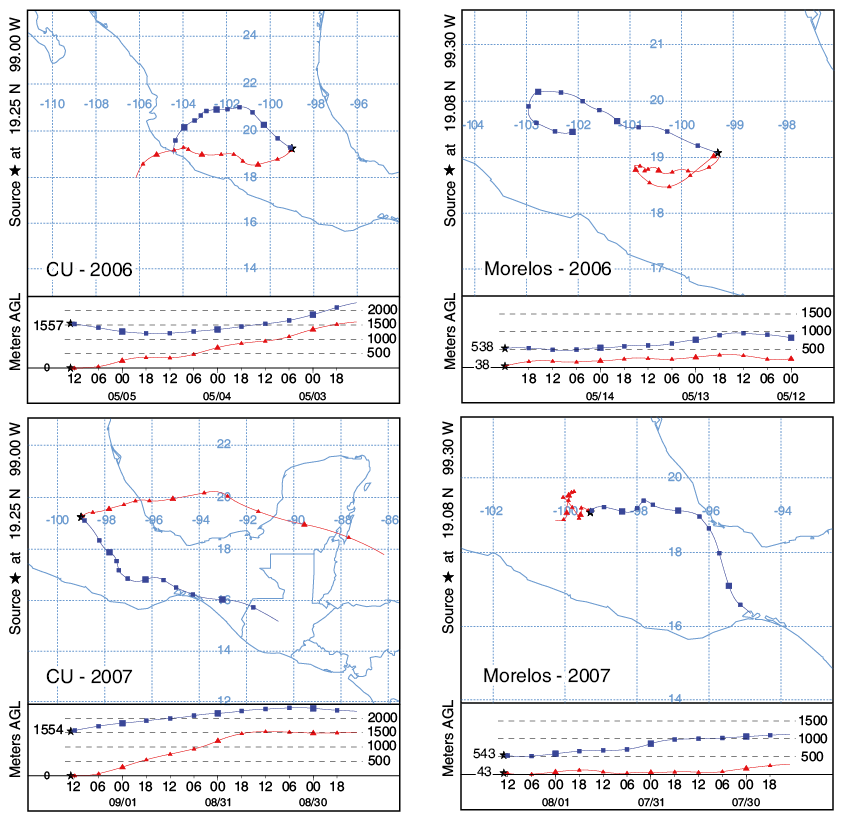
Air pollutant back trajectories were used to understand atmospheric transport and to identify the origins and pathways of air masses influencing the concentrations of the measured ions in rainwater.
Keywords: rainwater; ions; air pollution; back trajectories; Mexico.
RESUMEN
El objetivo de este trabajo fue el de evaluar la composición química del agua de lluvia en áreas urbanas y suburbanas del centro de México a fin de identificar las posibles fuentes de contaminantes en agua de lluvia. El agua de lluvia fue colectada en el Campus de la Universidad Nacional Autónoma de Mexico (Ciudad Universitaria, CU) al sur de la Ciudad de México y a 2200 m s.n.m. CU tiene muchas áreas verdes con tráfico moderado a intenso en donde la calidad del aire presenta serios problemas de contaminación por partículas. El otro sitio de muestreo es el área boscosa de Tlanepantla, Estado de Morelos, México, un área suburbana a 86 km al sur de la Ciudad de México. Un total de 145 muestras de agua de lluvia fueron colectadas en las temporadas de lluvia del 2006 al 2009. Los iones analizados fueron: SO42-, NO3-, Cl-, HCO3-, Na+, K+, Ca2+, Mg2+, NH4+ y H+. El amonio fue el catión más abundante en ambos sitios y es unos de los iones responsables de neutralizar compuestos ácidos en la atmósfera. La abundancia relativa de aniones inorgánicos presentes en el agua de lluvia se presenta en el siguiente orden: SO42- > NO3- > Cl-, para metales alcalinos el orden es: Ca2+ >Mg2+ > Na+ > K+, y Ca2+ >Mg2+ >K+ >Na+ para CU y Morelos, respectivamente. Un análisis de correlación muestra una fuerte correlación positiva entre los iones, lo cual indica que la fuente más probable es antropogénica. Las trayectorias de masas de aire fueron asociadas con las concentraciones de SO42-, Ca2+, Mg2+, NH4+ y H+ observadas durante cada día de lluvia. Se usaron cuatro factores en el análisis estadístico y a este se le dio un peso específico con cada factor. Los pesos específicos mayores de 0.5 son considerados como componentes significantes de cada factor. Los cuatro factores explican el 84.7 % de la variación total de los datos de la Ciudad de México y 66.9 % para Morelos. Todos los factores son asociados con los iones analizados. Las trayectorias de contaminantes fueron usadas para entender el transporte atmosférico e identificar origen y direcciones de las masas de aire que influyen las concentraciones de los iones medidos.
Palabras clave: agua de lluvia; iones; contaminación de aire; trayectorias de masas de aire; México.
Manuscript received: december 6, 2018
Corrected Manuscript received: may 17, 2019
Manuscript accepted: may 30, 2019
INTRODUCTION
Since the middle of the 20th century, studies of rainwater composition have been among the most important conducted by atmospheric scientists in the northern and southern Hemispheres. Rainwater plays an important role in cleaning the atmosphere, since by means of this process removes the greatest amount of contaminants present in it. The chemical composition of rainwater varies from one place to another and from one region to another, due to the influence of local sources, both natural and anthropogenic. Determining its chemical composition is important to understand the function of the transport of soluble components in the atmosphere, to understand the contribution of different sources of air pollution, and to establish the degree of environmental deterioration of a region. Therefore, the chemistry of wet deposits can provide insights into the temporal development of the atmosphere and its degree of contamination. Similarly, it can be used as an indicator in the evaluation of natural processes against anthropogenic influences (Niu et al., 2014). The chemistry of wet and dry deposits has continuously changed, as published papers have demonstrated (Aherne and Farrell, 2002; Baez et al., 1997. Studies of rainwater have been widely reported, with the purpose of knowing the degree of environmental deterioration of a site, mainly focused on the study of acid rain and alkaline dust neutralization mechanisms (Das et al., 2010). Therefore, the precipitation chemistry in both rural areas and urban areas has been subject to interesting studies in the last few decades (Galloway et al., 1983; Lee et al., 2000; Lacaux et al., 1992; Sanusi et al., 1996). In addition to the analysis of ion contents (NO3-, SO42-, NH4+, Cl-, Na+, Ca2+, Mg2+, K+) reported in this study, trace metals have been quantified due to their adverse environmental and human health effects and these results will be published elsewhere. Metals such as Pb, Cd, and Hg accumulate in the biosphere and may be toxic to living systems (Barrie et al., 1987; Galloway et al., 1982). Anthropogenic particles such as heavy metals are readily dissolved at the low pH values found in polluted rainwater (Migon et al., 1997); therefore, their bioavailability is enhanced. In Mexico City, the chemical composition of rainwater has been continuously studied due to the heavy air pollution that characterizes the metropolitan area of Mexico City. So, rainwater composition is a very important way to monitor the scavenging atmospheric processes of particulate matter and gases in the area (Liu et al., 2012). The purpose of the study was to evaluate the chemical composition of rainwater in urban and suburban areas of central Mexico and to identify the possible sources of rainwater contamination.
Sampling sites
The urban site was located at the Centro de Ciencias de la Atmósfera (CCA) located at 19°19’34” N latitude and 99°10’55” W longitude inside the Universidad Nacional Autónoma de México, campus Ciudad Universitaria (CU), in the southern part of Mexico City at 2200 m a.s.l. The University has many green areas with high to moderate traffic densities. The other sampling site is a forested rural area (Tlalnepantla), State of Morelos, Mexico, located at 19°00’28” N latitude and 98°59´51” W longitude in the southern foothills of the Eje Neovolcánico at 2060 m a.s.l. Tlalnepantla borders on the north with the metropolitan area of Mexico City and the State of Mexico, on the southeast with Totolapan, the west with Tepoztlán, the south with Tlayacapan, and Cuautla City is further to the south and has moderate human activity. Pine and caducifolious forests lie to northeast and the northwest of the sampling site, with light vehicular traffic (Figure 1).
A total of 145 rainwater samples were collected using automatic wet/dry precipitation collectors (Andersen, General Metal Works, Inc.) for inorganic ions on the weekends from mid-May to the end of October, which is the rainy season in central Mexico, during the 2006 to 2009 period. The samples were collected in standard high-density polyethylene (HDPE) buckets. To minimize contamination, the collector lid arms were Teflon-coated and the original aluminum cover and plastic-covered pad were replaced with a Teflon lid and pad. In the rural area Tlalnepantla, the sampling was done in passive collectors that consisted of a funnel attached to a high-density polyethylene (HDPE) bottle supported by a rod at a height of 1.5 m above ground level (Galloway et al., 1983). After the rainy events, the collection buckets were tightly sealed with polyethylene covers and were sent to the chemical laboratory of the CCA. At the laboratory, all collected samples were filtered through 0.40 µm polycarbonate membrane filters, which were leached with 300 ml of deionized water. For the ion analysis, samples were poured into HDPE bottles, capped, and kept at 4 °C until analysis. The buckets used to collect the rain samples for major ion analysis and the flask and storage bottles were previously washed, brushed, and rinsed with distilled water and then deionized water (DI). The sample was treated under a clean hood (EPA) maintained at positive pressure to limit the introduction of contaminants from the laboratory.